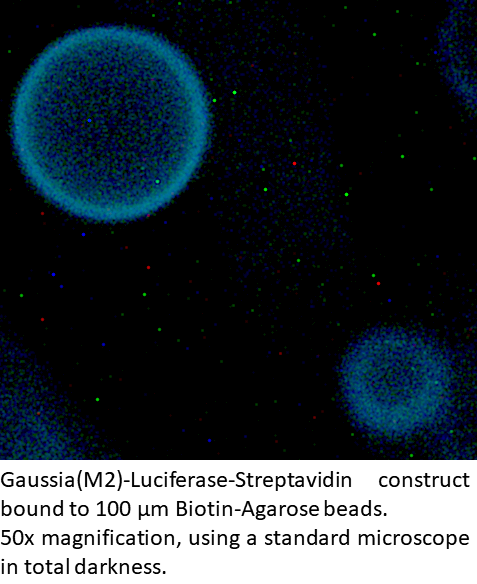
Gaussia(M2)-Luciferase-Streptavidin is a conjugate of one of the brightest luciferases and a protein with the highest known affinity in nature. This fusion protein can be used to detect trace amounts of biotinylated protein, DNA, or other biotinylated bio-molecules.
- Details & Info
- Protocols & Data
- Related Products